- Visibility 398 Views
- Downloads 59 Downloads
- Permissions
- DOI 10.18231/j.ijpo.2021.025
-
CrossMark
- Citation
Spectrum of ovarian tumors in a tertiary care hospital
- Author Details:
-
Priyanka D Dhende *
-
Lalita Y Patil
-
Kusum Jashnani
Abstract
Background: Ovarian tumors are heterogeneous group of neoplasms with wide range of histopathological patterns. It is one of the leading causes of mortality in females. Often difficult to detect until they are advanced in stage or increased in size as symptoms are vague and insidious. Treatment and prognosis is based upon accurate surgical staging and thorough pathological evaluation.
Materials and Methods: A total 150 cases were studied from June 2014 to May 2017. Their age, clinical presentation and histopathological findings were reviewed and analyzed.
Results: Out of 150 cases of ovarian tumors studied 119(79.3%) were benign, 9 (6.0%) borderline and 22 (14.6%) malignant. Age ranged from 9 to 80 years. The commonest presenting complaint in malignant and benign cases was pain in abdomen and in borderline cases abdominal distension. CA125, CEA and LDH were most commonly raised in 53.8%, 23% and 100% malignant cases respectively. Surface epithelial tumors were 68% followed by germ cell tumors 28% and sex cord stromal tumors 4%.
Introduction
Ovarian tumors are heterogeneous group of neoplasm with wide range of histological patterns. It accounts for 3% of all cancers in female and is the 5th leading cause of death due to cancer in United States.[1] Ovarian tumors are often difficult to detect until they are advanced in size or stage. Hence it is often called as “silent killer”. The treatment and prognosis of the ovarian tumors is based upon accurate surgical staging and a thorough pathological evaluation.
The need of reliable specific serum biomarkers for early detection of ovarian tumors remains a long awaited priority. In recent years there is significant development in use of immunohistochemistry for diagnosing ovarian tumors. There are distinctive immunohistochemical features for each of the three main categories of ovarian tumors.
There is increasing trend of ovarian carcinoma in females. The application of current knowledge and techniques would revolutionize the ovarian cancer statistics. This will help large number of women in each year and lead towards the more satisfactory therapy of disease.
Materials and Methods
This is a retrospective observational study of 3 years duration from 1st June 2014 to 30th May 2017 during which 150 cases were studied. Their clinical data (age, site, clinical presentation and levels of tumor markers), radiologic findings and gross appearance were obtained from the surgical histopathological record section of the institute. Formalin fixed paraffin embedded tissue sections stained with Hematoxylin and Eosin was retrieved and reviewed. Immunohistochemistry was performed in borderline and malignant cases.
Results
Total 150 cases of ovarian tumors were studied of which the most common was benign i.e.119 (79.3%) cases followed by 22 (14.6%) malignant cases and 9 (6.0%) borderline cases.
|
Type |
No. of cases |
Percentage |
|
Surface epithelial tumors |
102 |
68% |
|
Germ cell tumors |
42 |
28% |
|
Sex-cord stromal tumors |
6 |
4% |
Age distribution
In present study, the women affected were in the age range of 9 to 80 years with the mean of 38.6 years. Overall, the peak was seen at 31-40 years of age. The benign ovarian tumors were most common in the age group of 31-40yrs, borderline 41 -60yrs and malignant 11-20yrs.
The youngest patient, 9yrs old female child was diagnosed as Yolk sac tumor of the ovary and the eldest patient, 80yrs old female was diagnosed as benign mixed epithelial tumor (benign mucinous cystadenoma with Brenner component in mural nodule).
Clinical presentation
The most common presentation in benign and malignant ovarian tumors was abdominal pain seen in 76 (64.4%) and 10(45.4%) cases respectively whereas most common presentation in borderline ovarian tumors was abdominal distension in 4 (44.4%) cases.
Out of 150 ovarian tumors, 7 (4.6%) cases detected incidentally out of which 6 women detected at the time of routine antenatal ultrasonography checkup and one detected at radiological workup for cholelithiasis. Out of these 7 incidentally detected cases 6 were benign tumors and 1 was borderline serous tumor. Of these 6 benign tumors 4 were benign serous cystadenoma, 1 benign mucinous cystadenoma and1 was mature cystic teratoma.
Tumor markers
CA125, CEA and LDH were raised most commonly in malignant cases. The highest value of CA125 in present study was 2075.7u/ml which was seen in 1 case of malignant serous cystadenocarcinoma. CEA was markedly raised in 2 cases of mucinous cystadenocarcinoma and 1 case of yolk sac tumor. AFP was raised with levels more than 10,000 in 4 cases out of which 3 cases were Yolk sac tumors and 1 case was immature teratoma.
Radiological finding
Radiological findings were available in 102 (85.7%) out of 119 cases of benign ovarian tumors and 9 (100%) cases of borderline and 22 (100%) malignant ovarian tumors. The most common radiologic finding was cystic lesion seen in 93 (91.1%) benign cases and 5 (55.5%) borderline cases respectively. In malignant ovarian tumors the commonest radiologic finding were heterogeneous solid-cystic mass lesion with contrast enhancement seen in 14 (13.6%) cases.
Distribution of ovarian tumors
Among benign ovarian tumors serous cystadenoma was the commonest followed by mature cystic teratoma. The commonest borderline ovarian tumor was Borderline mucinous tumor. There was 1 case of Borderline Micropapillary serous tumor.
|
|
Surface epithelial tumors |
Germ cell tumors |
Sex cord stromal tumor |
||||
|
Distribution |
Serous |
Mucinous |
Mixed epithelial |
Mature cystic teratoma |
Struma ovari |
Fibroma |
Fibro-thecoma |
|
No. of cases n=119 |
47 |
31 |
5 |
33 |
1 |
1 |
1 |
|
Percentage |
39.4% |
26.0% |
4.2% |
27.7% |
0.8% |
0.8% |
0.8% |
|
Distribution |
Borderline Serous |
Borderline mucinous |
Borderline endometrioid |
|
No. of cases (n=9) |
3 |
5 |
1 |
|
Percentage |
33.3% |
55.5% |
11.1% |
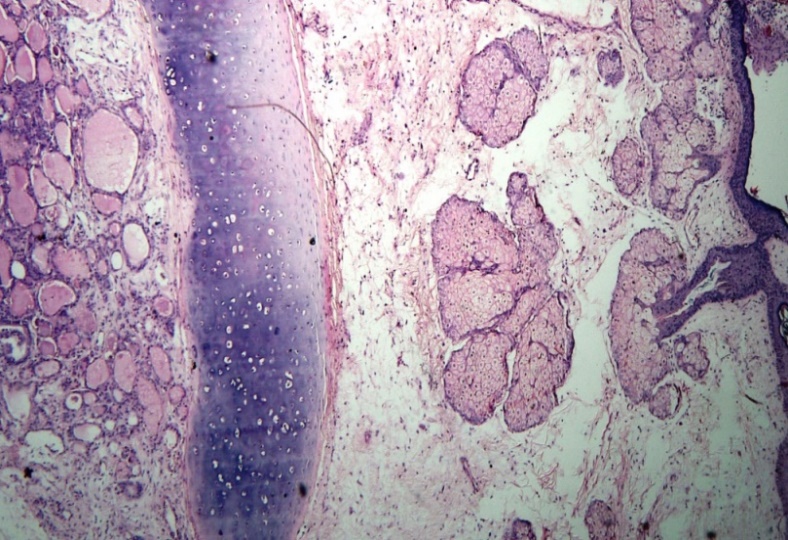
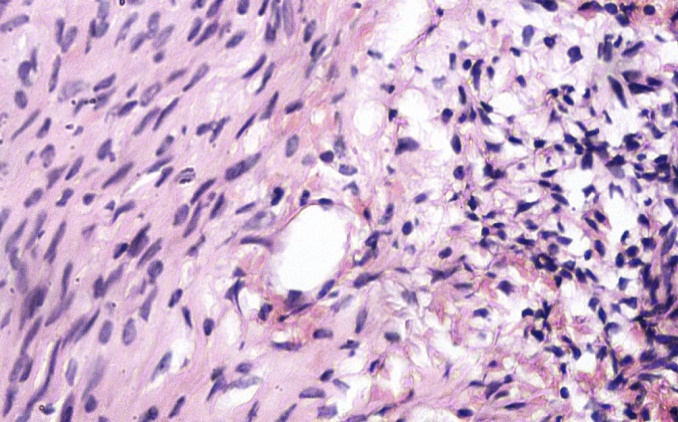
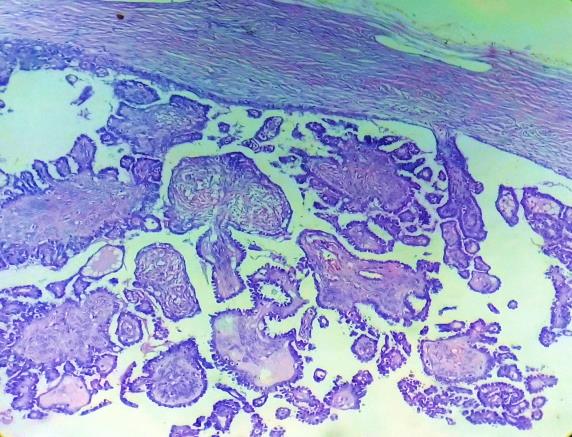
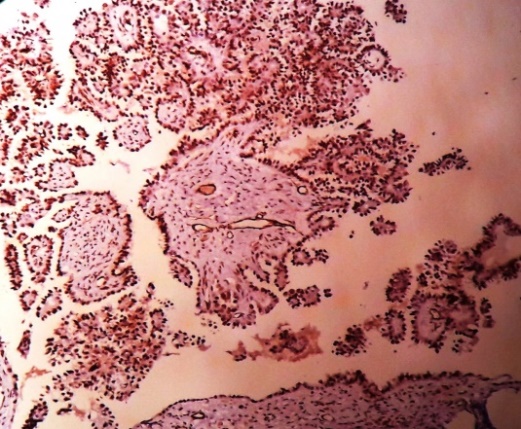
|
Malignant tumors |
No. of cases |
Percentage |
|
Serous carcinoma |
1 |
4.5% |
|
Mucinous carcinoma |
7 |
31.8% |
|
Borderline mucinous with neuroendocrine carcinoma component |
1 |
4.5% |
|
Granulosa cell tumor |
3 |
18.6% |
|
Sertoli cell tumor |
1 |
4.5% |
|
Immature teratoma |
1 |
4.5% |
|
Teratoma with poorly differentiated component |
1 |
4.5% |
|
Yolk sac tumor (YST) |
2 |
9.0% |
|
Dysgerminoma (Dys) |
3 |
18.6% |
|
YST+Dys |
1 |
4.5% |
|
Metastasis |
1 |
4.5% |
The most common category of malignant ovarian tumor was surface epithelial tumor seen in 40.9% cases followed by germ cell tumors seen in 36.36% cases. The most common malignant ovarian tumor was primary mucinous carcinoma seen in 7 (31.8%) cases out of 22 malignant ovarian tumors.
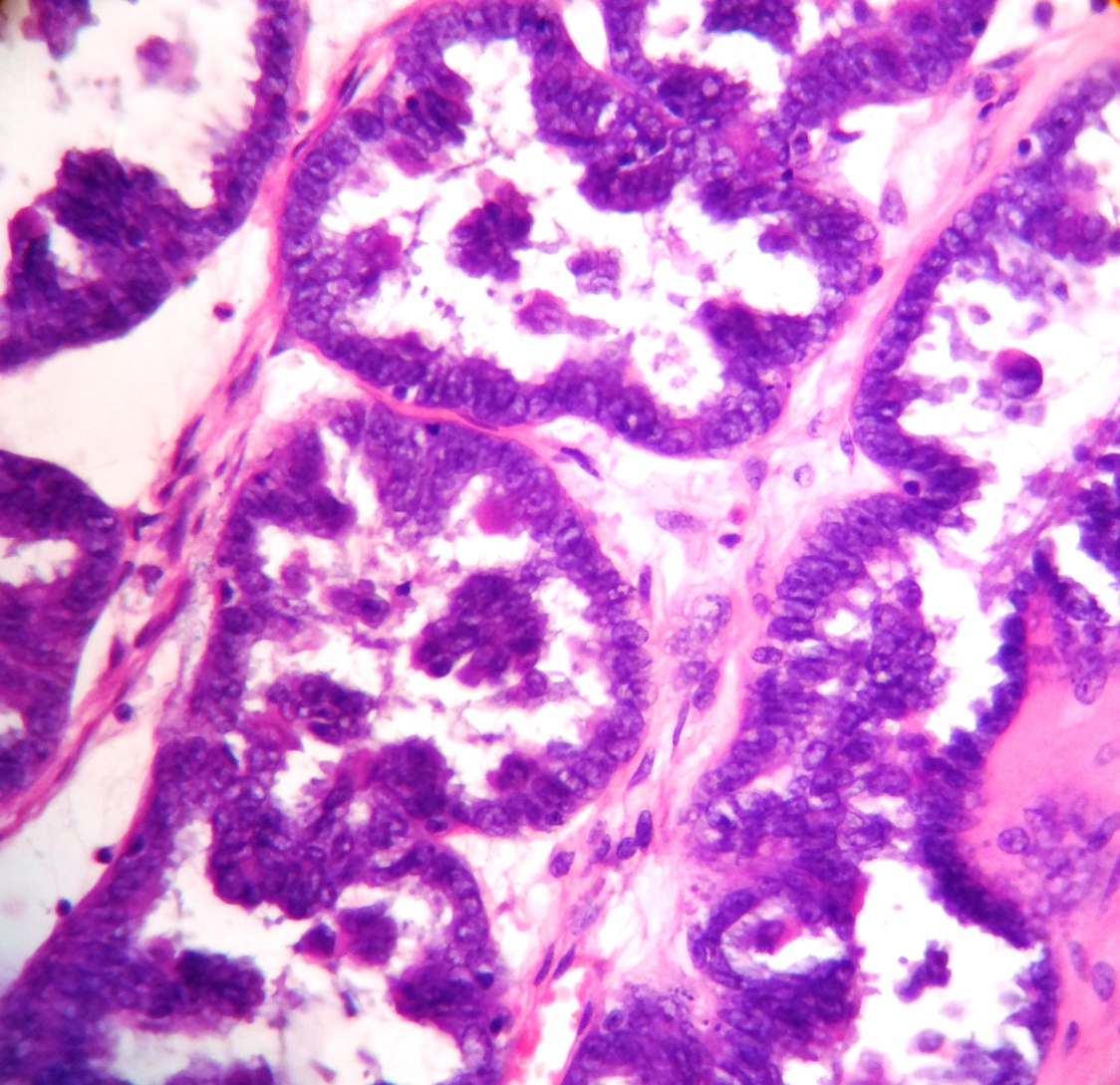
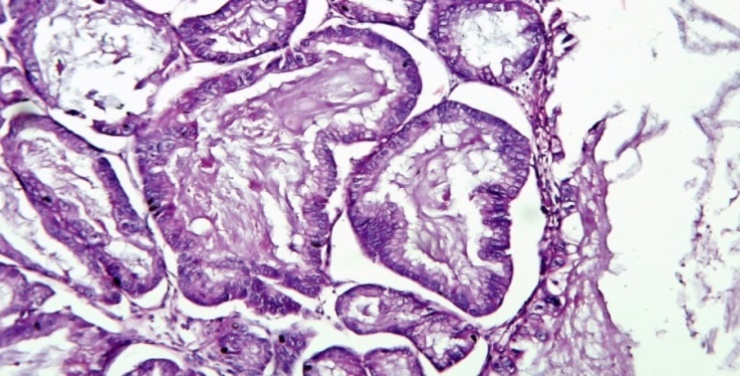
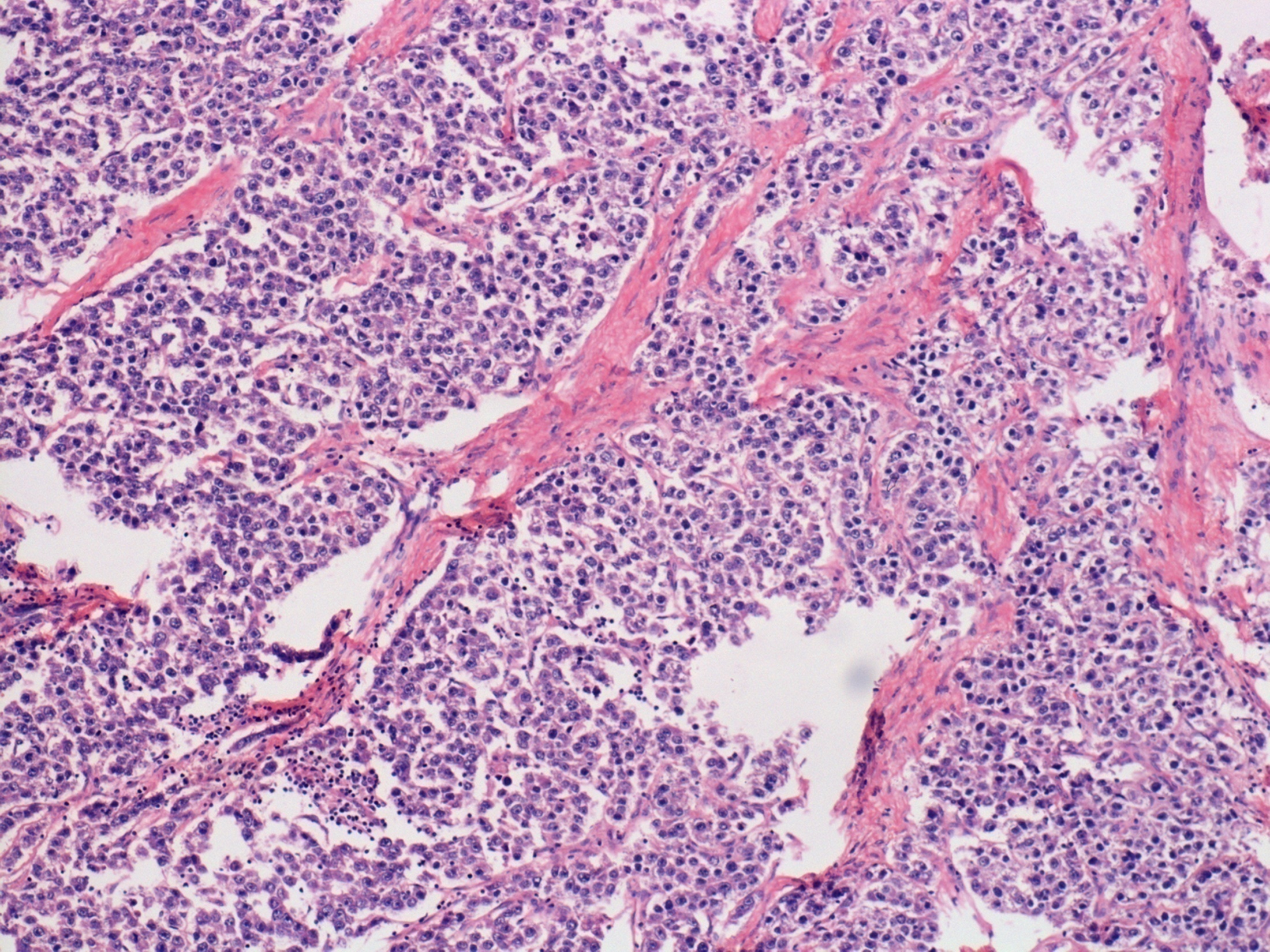
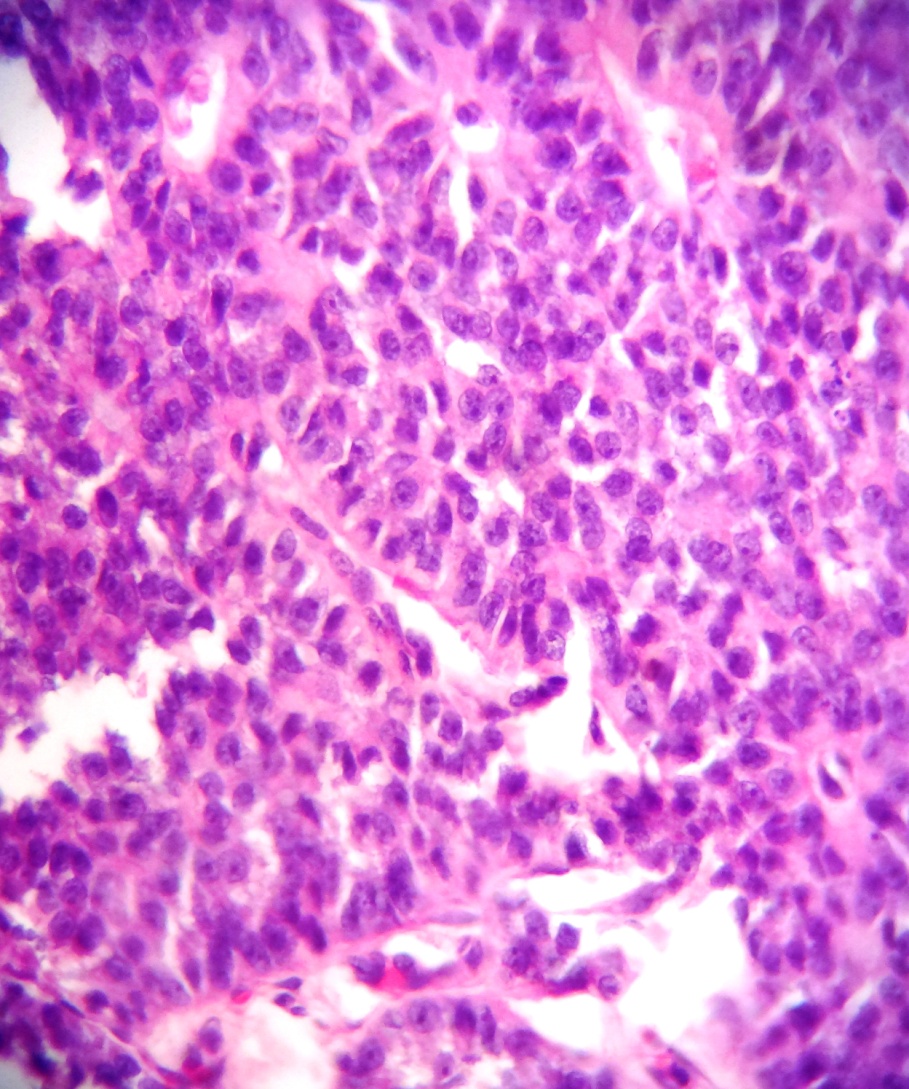
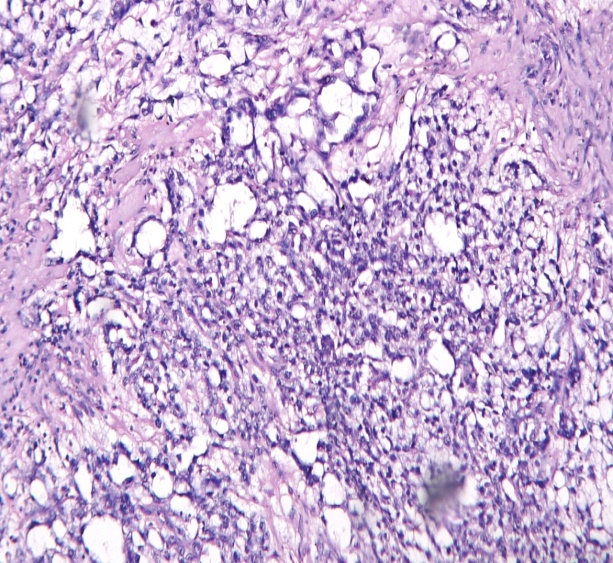
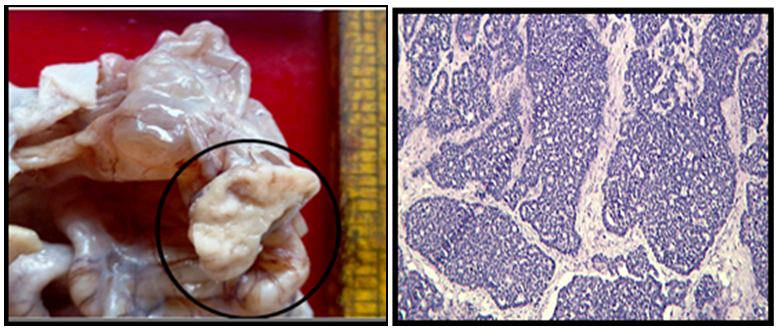
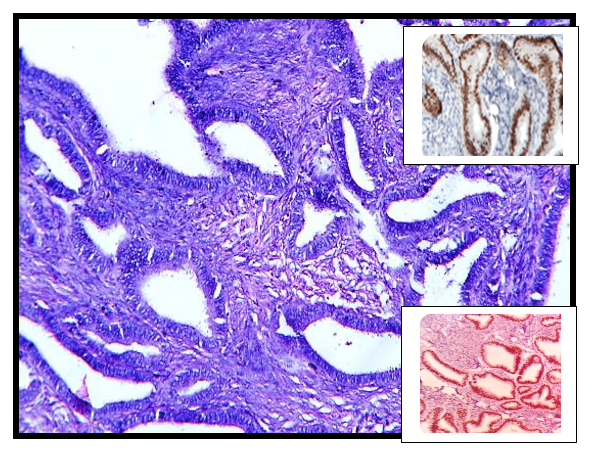
Size distribution
In present study the smallest tumor was of size 2.5cm in diameter which was benign serous cystadenoma and the largest was of 30cm in diameter which was benign mucinous cystadenoma. The benign tumors size were ranging from 2.5 to 30cm with most common in the range of 6-10cm whereas borderline tumors were ranging from 5-29cm with most common in 16-20cm range and malignant tumors were ranging from 5 to 22cm with most common in range of 11-20cm.
Laterality
Out of total 150 cases of ovarian tumors 142 (94.6%) cases were unilateral and 8 (5.3%) cases were bilateral. Out of 8 cases of bilateral ovarian tumors 6 cases were benign serous cystadenoma and 1 case each of primary mucinous cystadenocarcinoma and secondary mucinous carcinoma (metastatic).
Consistency
Of all benign and borderline ovarian tumors 112 cases (94.1%) of benign and 7 cases (77.7%) of borderline were cystic in nature. Among 22 cases of malignant ovarian tumors 11 cases (50%) were solid and 7 cases (31.8%) were solid-cystic in nature.
Immunohistochemistry
|
IHC |
Serous tumors |
Mucinous tumors |
||||||
|
WT1 N=4 |
PAX8 N=3 |
P53 N=1 |
CK7 N=13 |
CK20 N=13 |
CDX2 N=5 |
WT1 N=3 |
B-catenin N=3 |
|
|
Borderline |
2(66.6%) |
3(100%) |
- |
5(100%) |
1(20%) |
- |
- |
- |
|
Malignant |
1 |
- |
1 |
8(100%) |
1(12.5% |
0 |
2(66.6%) |
1 |
WT1 positivity was noted in 3 (75%) out of total 4 serous tumors, CK 7 positivity in all 13 (100%) cases of mucinous tumors whereas CK20 positivity in 2 (15.3%) cases of mucinous tumors. Calretinin positivity was noted in 2 (66.6%) of 3 cases of Granulosa cell tumors whereas inhibin positivity in 1 (33.3%) case out of 3 cases of Granulosa cell tumor. In all 3 cases of dysgerminoma PLAP positivity was noted and in 2 cases of yolk sac tumor AFP positive was seen. In Sertoli cell tumor vimentin was strongly and diffusely positive with EMA and inhibin negativity.
Discussion
In this study of 150 ovarian tumors, surface epithelial tumors were 101 (67.3%) cases followed by 42 (28.0%) cases of germ cell tumors and 6 (4.0%) cases of sex cord stromal tumors. Surface epithelial tumors were the commonest encountered tumors in present study similar to Agarwal et al[2] and Bhagyalaxmi et al.[3] The percentage of germ cell tumors was higher as compared to Mondal et al,[4] Agarwal et al[2] and Bhagyalaxmi et al.[3] Surprisingly in study of Vaidya et al,[4] the percentage of germ cell tumor was very high i.e. 51.5%.
|
Categories |
Present study (2014-1=2017) n=150 |
Bhagyalaxmi et al [3] (2011-2014) n=267 |
Agarwal et al [2](2006-2011) n=226 |
Mondal et al [4] (2001-2010) n=957 |
Vaidya et al[5] (2011-2013)n=363 |
|
Surface epithelial tumors |
67.3% |
80% |
72.1% |
67.9% |
43.5% |
|
Germ cell tumors |
28% |
14.2% |
19.2% |
23.1% |
51.5% |
|
Sex-cord stromal tumors |
4.0% |
4.1% |
7.1% |
5.6% |
3.3% |
|
|
Present study(2014-2017) |
Agarwal et al [2] (2006-2011) |
Mondal et al [4] (2001-2010) |
Bhagyalaxmi A et al [3] (2011-2014) |
|
Total |
150 |
226 |
957 |
26 |
|
Benign |
79.3% |
61.1% |
63.1% |
78.3% |
|
Borderline |
6% |
7.1% |
7.3% |
3.7% |
|
Malignant |
14.6% |
31.9% |
29.6% |
18% |
The frequency of malignant tumors in present study was 14.6% which is lower than frequencies of the same in studies of Agarwal et al,[2] Mondal et al[4] and Bhagyalaxmi et al[3] which is a good sign.
The most common benign ovarian tumor in present study was benign serous cystadenoma seen in 39.4% cases which is comparable to the studies of Agarwal et al,[2] Bhagyalaxmi et al[3] and Mondal et al.[4] The second most common benign ovarian tumor in present study was mature cystic teratoma which is similar to the study of Mondal et al[4] but is in discordance with Agarwal et al [2] and Bhagyalaxmi et al[3] in which benign mucinous cystadenoma was second most common.
The most common borderline ovarian tumor in present study is borderline mucinous tumor i.e. 55.5% which is similar to Agarwal et al[2] and Bhagyalaxmi et al[3] but in study of Mondal et al [4] borderline serous tumor was most common.
In studies conducted by Agarwal et al, [2] Bhagyalaxmi et al[3] and Mondal et al[4] serous cystadenocarcinoma was most common tumor among malignant ovarian tumors in contrary to this in present study mucinous carcinoma was most common.
In present study abdominal pain was the most common presentation in 77.2% malignant cases. This finding is very well similar to which is comparable with Wasim et al,[6] Mankar et al[7] and Amreem khan et al.[8]
|
Tumor marker |
Present study (2014-2017) |
Chen et al[9] (2017) |
Agarwal et al[2] (2006-2011) |
|
|
CA 125 |
Sensitivity |
58.3% |
62.75% |
90% |
|
Specificity |
90.0% |
70.6% |
40% |
|
|
CEA |
Sensitivity |
23.0% |
35.7% |
- |
|
Specificity |
95.0% |
79% |
- |
In present study the sensitivity and specificity of CA125 and CEA was comparable to the study conducted by Chen et al[9] however, it was not similar with the study of Agarwal et al.
The AFP was 100% specific for yolk sac tumor.
In present study 94.6% cases of the ovarian tumors were unilateral and 5.3% cases were bilateral. The most common among bilateral tumors were benign serous cystadenoma. However, in studies of Mondal et al[4] and Bhagyalaxmi et al[3] malignant serous carcinomas were most common among the bilateral tumors.
In present study right side preponderance was seen which is similar to Pilli et al.[10] There was no side (right or left) preponderance seen in study of Agarwal et al.[2]
It can be seen from present study that the size is not an important factor in assessing the nature of tumor similar to Agarwal et al[2] as size range in benign, borderline and malignant tumors were along similar size.
We encountered a rare case of mature cystic teratoma with foci of poorly differentiated carcinoma in a 72 years old female. This tumor was of size 20cm, solid cystic in nature. Serum CA19.9 was markedly raised. IHC marker pan CK7 was positive, CK20, WT1, PAX-8, synaptophysin were negative. The incidence of malignancy arising in a mature cystic teratoma is 1-2% as per Chaudhry et al.[11] Another rare case of borderline mucinous tumor with neuroendocrine carcinoma component was encountered in this study.
Overall ovarian tumors were found in the age range of 9–80 years in the present studies. Maximum number of cases overall were in the age range of 31–40 years. Benign ovarian tumors were most common in the age group of 31-40 years and borderline ovarian tumors in 41-60 years which is similar to Bhagyalaxmi et al,[3] Mondal et al[4] and Agarwal et al.[2]
In present study the commonest age group affected in malignant cases was 11-20 years which is in discordance with other studies in literature but it is comparable with Vaidya et al[5] in which 21-30 years was commonest. This could be due to higher number of germ cell tumors.
|
|
Present study (2014-2017) |
Kriplani and Patel et al[12] (2009-2011) |
Cathro et al[13] (2002) |
|
|
Mucinous tumors |
Ck7 |
n=13 13 (100%) positive |
n=5 4(80%) |
n=14 12(85.7%) |
|
Ck20 |
2 (15.3%) positive |
2(50%) |
6(42.8%) |
|
|
Serous tumors |
WT1 |
n=4 3 (75%) |
n=22 18(81.8%) |
- |
CK7 showed strong and diffuse cytoplasmic positivity in 100% primary mucinous tumors which is comparable with Kriplani and Patel et al[12] and Cathro et al[13] for diagnosing primary mucinous tumors. CK7 and CK20 both were positive in two cases of mucinous tumor and in view of morphology and IHC like CDX2 and B-Catenin diagnosis of primary mucinous tumor was favored.
WT1 showed cytoplasmic positivity in 75% cases of serous tumors which is similar to study of Kriplani and Patel et al.[12]
Conclusion
The present study was undertaken to evaluate the various histopathological spectrum of ovarian tumors, their commonest age group distribution and histopathological and clinical features correlation. Ovarian tumors exhibit wide range of histopathological spectrum in our institute. The percentage of surface epithelial tumors constituted the greatest proportion followed by germ cell tumors. Serous cystadenoma was commonest followed by mature cystic teratoma in benign cases. Borderline mucinous cystadenoma was commonest in borderline. In malignant cases mucinous cystadenocarcinoma was commonest. Overall middle age is commonly affected. Patients commonly presents with pain in abdomen. Radiology has less specificity and but is complementary for diagnosing few malignant cases. Biochemical markers were contributory to distinguish benign and malignant cases. The size of the tumor is not related to the nature of the tumor but presence solid elements makes malignancy more likely.
Common problems encountered while diagnosing tumors of epithelial origin especially serous and mucinous tumors is that tumor may include benign and borderline components in few areas and malignant counterparts in other areas. Hence, extensive sampling and sampling from solid areas as well as from mural nodules is very important. Also, many times, it is difficult to differentiate primary mucinous carcinoma from metastasis especially from appendix, intestine stomach, cervix, pancreas etc. in such situation IHC especially CK7, CK20, CDX2 and B- catenin along with clinical details plays important role.
Source of Funding
None.
Conflict of Interest
None.
References
- Ellenson L, Pirog E. The Female Genital Tract. Robbins and Cotran Pathologic Basis of Disease. 2010. [Google Scholar]
- Agrawal P, Kulkarni D, Chakrabarti P, Chourasia S, Dixit M, Gupta K. Clinicopathological spectrum of ovarian tumors: A 5-year experience in a tertiary health care center. J Basic Clin Reprod Sci. 2015;4(2):90-6. [Google Scholar] [Crossref]
- Bhagyalakshmi A, Sreelekha A, Sridevi S, Chandralekha J, Parvathi G, Venkatalakshmi A. Prospective study of histopathological patterns of ovarian tumours in a tertiary care centre. Int J Res Med Sci. 2014;2(2):448-56. [Google Scholar] [Crossref]
- Mondal S, Nag D, Mondal P, Banyopadhyay R, Roychowdhury S, Sinha S. Histologic pattern, bilaterality and clinical evaluation of 957 ovarian neoplasms: A 10-year study in a tertiary hospital of eastern India. J Cancer Rese Ther. 2011;7(4):433-7. [Google Scholar] [Crossref]
- Vaidya S, Sharma P, KC S, Vaidya S. Spectrum of ovarian tumors in a referral hospital in Nepal. J Pathol Nepal. 2014;4(7):539-43. [Google Scholar] [Crossref]
- Wasim T. P1118 Comparison of clinical presentation of benign and malignant ovarian tumors. Int J Gynecol Obstet. 2009;107:S725-6. [Google Scholar] [Crossref]
- Mankar D, Jain G. Histopathological profile of ovarian tumours: A twelve year institutional experience. Muller J Med Sci Res. 2015;6(2):107-11. [Google Scholar] [Crossref]
- Ambareen K, Khusro S. Presenting signs and symptoms of ovarian cancer at a tertiary care hospital. J Pak Med Assoc. 2010;60(4):260-2. [Google Scholar]
- Chen F, Shen J, Wang J, Cai P, Huang Y. Clinical analysis of four serum tumor markers in 458 patients with ovarian tumors: diagnostic value of the combined use of HE4, CA125, CA19-9, and CEA in ovarian tumors. Cancer Manag Res. 2018;10:1313-8. [Google Scholar]
- Pilli G, Suneeta K, Dhaded A, Yenni V. Ovarian tumors: A study of 282 cases. J Indian Med Assoc. 2002;100(420):423-7. [Google Scholar]
- Chaudhry S, Hussain R. Squamous cell carcinoma arising in mature cystic teratoma (dermoid cyst)-a rare presentation. J Pak Med Assoc. 2013;63(4):521-3. [Google Scholar]
- Cathro H, Stoler M. Expression of Cytokeratins 7 and 20 in Ovarian Neoplasia. Am J Clin Pathol. 2002;117(6):944-51. [Google Scholar] [Crossref]
- Kriplani D, Patel M. Immunohistochemistry: A diagnostic aid in differentiating primary epithelial ovarian tumors and tumors metastatic to the ovary. South Asian J Cancer. 2013;02(04):254-8. [Google Scholar] [Crossref]
How to Cite This Article
Vancouver
Dhende PD, Patil LY, Jashnani K. Spectrum of ovarian tumors in a tertiary care hospital [Internet]. Indian J Pathol Oncol. 2021 [cited 2025 Oct 25];8(1):133-139. Available from: https://doi.org/10.18231/j.ijpo.2021.025
APA
Dhende, P. D., Patil, L. Y., Jashnani, K. (2021). Spectrum of ovarian tumors in a tertiary care hospital. Indian J Pathol Oncol, 8(1), 133-139. https://doi.org/10.18231/j.ijpo.2021.025
MLA
Dhende, Priyanka D, Patil, Lalita Y, Jashnani, Kusum. "Spectrum of ovarian tumors in a tertiary care hospital." Indian J Pathol Oncol, vol. 8, no. 1, 2021, pp. 133-139. https://doi.org/10.18231/j.ijpo.2021.025
Chicago
Dhende, P. D., Patil, L. Y., Jashnani, K.. "Spectrum of ovarian tumors in a tertiary care hospital." Indian J Pathol Oncol 8, no. 1 (2021): 133-139. https://doi.org/10.18231/j.ijpo.2021.025
